
MDS
Advertisement
The primary end point for RBC transfusion independence was not met but the cohort still had significant anemia improvement.
The phase 3 VERONA trial, which compared this combination with placebo plus azacitidine, observed no new safety signals.
The study will assess the overall response to olutasidenib plus azacitidine in higher-risk MDS, CMML, and advanced MPNs.
Reactive oxygen species levels decreased and patients with evident bone marrow failure showed hematological response.
A study has explored the clinical impact of both mutation-based and non-mutation-based p53 dysfunction in MDS.
Preclinical models show significant efficacy in combos against AML cell lines with FLT3, KMT2A, NPM1, and TP53 mutations.
Mice treated with the novel approach had a “significantly lower tumor burden” compared with mice that received controls.
The agent recalibrates the immune system to attack tumors and increases cancer susceptibility to standard of care therapy.
Investigational T-Cell Immunotherapy Superior to Standard HSCT in Rate of Survival Plus GvHD Freedom
A phase 3 study comparing the treatments in multiple hematologic malignancies also saw greater overall survival at one year.Imetelstat is indicated for patients with lower-risk MDS who had an unsatisfactory response to or are ineligible to ESAs.
Detectable MRD before HSCT is associated with worse survival in patients with MDS/MPN compared with undetectable MRD.
Overall survival, nonrelapse mortality, and relapse rates were also assessed in the phase 2 trial which had these results.
The new indication is for use of the alkylating agent with fludarabine in a preparatory combination for allogeneic HSCT.
Juan Jose Rodriguez-Sevilla, MD, PhD, summarizes current clinical knowledge about inflammation within MDS pathobiology.
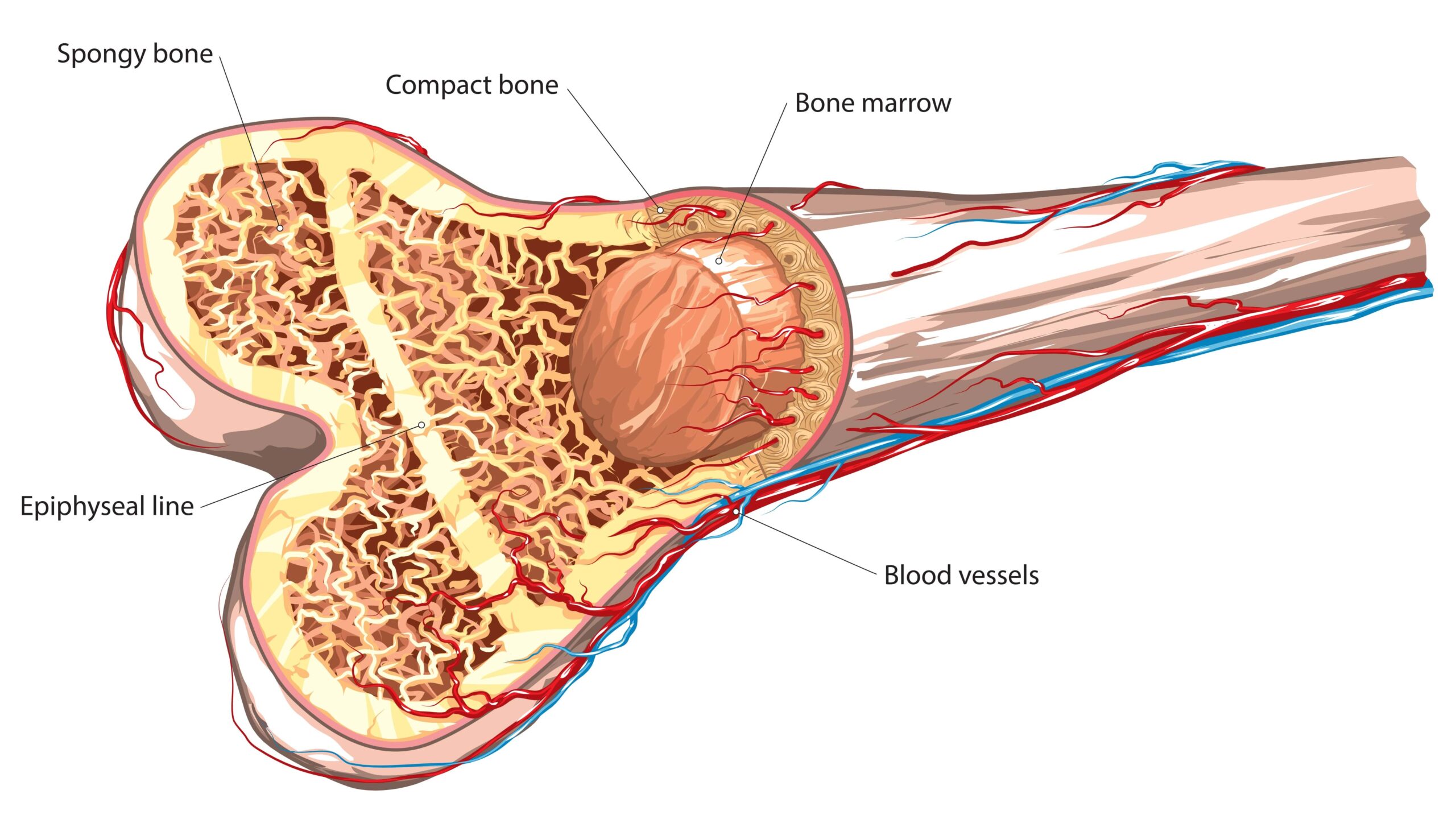
An MDS HSPC signature generated from the model presents possible targets for treatment in patients with low-risk disease.
The study's results especially highlight potential aberrant gene regulation by ZMAT2 and SMARCD3.
The study showed a mortality risk reduction benefit in certain mutations while for others larger studies are needed.
Researchers noted that confirmation of this relationship in lower-risk disease would require a larger study.
Researchers argue from their findings that the 2023 criteria more accurately present the performance of ivosidenib in MDS.
The combination in a phase II trial produced high complete response rates in patients with newly diagnosed AML.
Advertisement










 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.