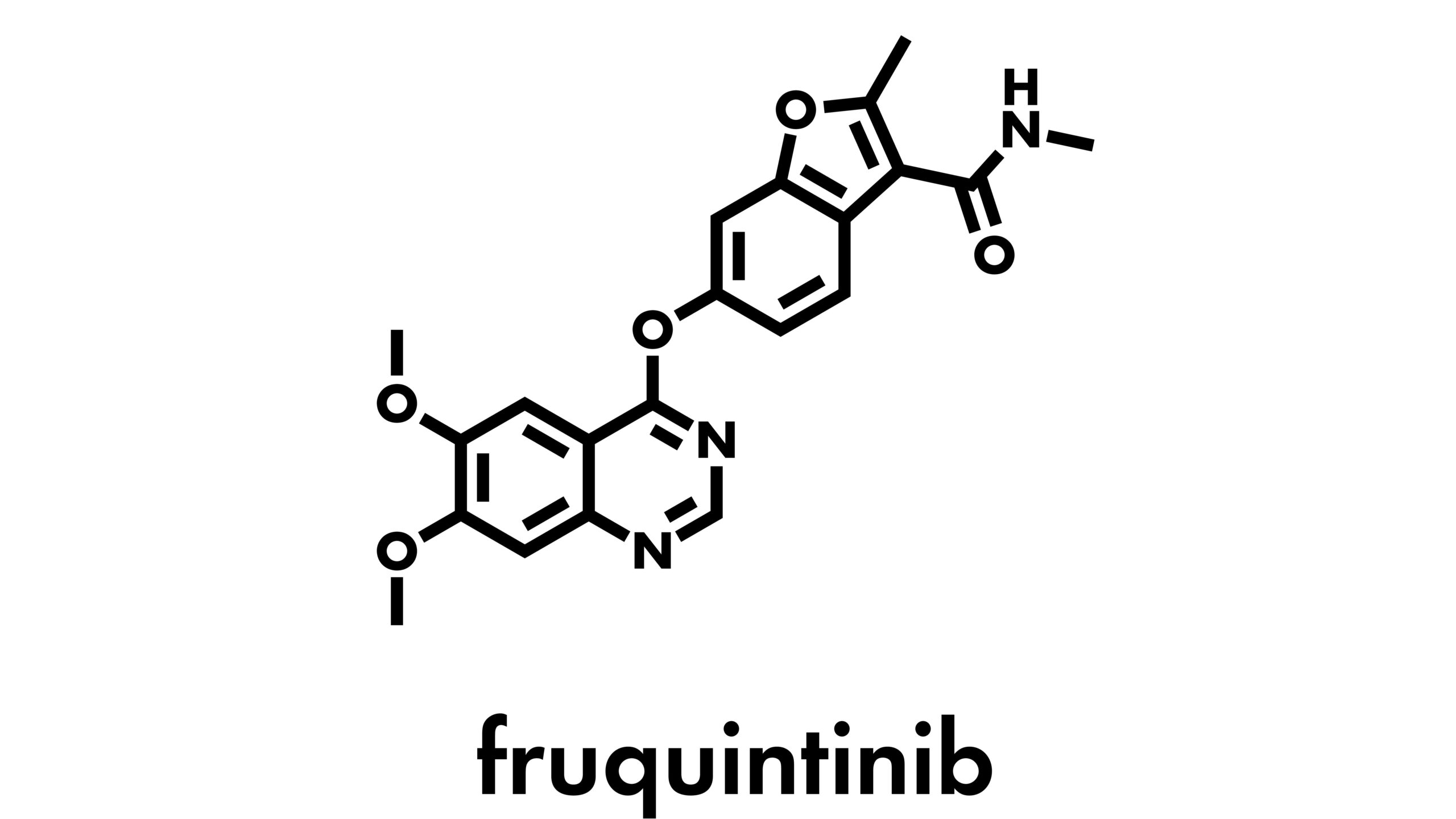
Researchers, led by Arvind Dasari, noted that data on effective treatment options for patients with advanced, chemotherapy-refractory colorectal cancer are sparse. They evaluated the safety and efficacy of fruquintinib, a potent, selective oral vascular endothelial growth factor receptors (VEGFRs) inhibitor.
Based on their phase 3 trial results, the authors reported that fruquintinib significantly improved overall survival (OS) compared with placebo in patients with refractory metastatic colorectal cancer.
“These data support the use of fruquintinib as a global treatment option for patients with refractory metastatic colorectal cancer,” they suggested. The study’s findings were presented in Lancet.
Fruquintinib Promising for Advanced Colorectal Cancer
The phase 3 FRESCO-2 trial was performed at 124 centers adcross 14 countries from August 2020 to December 2021. All participants had metastatic colorectal adenocarcinomas and had progressed or failed on standard cytotoxic and targeted therapies.
A total of 691 patients were assigned to receive either oral fruquintinib 5 mg (n=461) or placebo (n=230) once daily from day 1 to day 21 during 28-day cycles. The cohort had a median of 4 prior lines of systemic therapy (interquartile range, 3-6), with 73% (n=502) having received more than 3 lines. The primary end point was OS, defined as time from randomization to death from any cause. The final analysis included 480 events.
According to the report, the median OS in the fruquintinib group was 7.4 months (95% CI, 6.7-8.2) compared with 4.8 months (95% CI, 4.0-5.8) in the placebo group (hazard ratio, 0.66; 95% CI, 0.55-0.80; P<.0001).
Researchers noted grade 3 or higher adverse events oc cured in 286 (63%) out of 456 patients in the fruquintinib group compared with 116 (50%) out of 230 patients in the placebo group. The most common grade 3 or higher events in the fruquintinib group were hypertension, asthenia, and hand-foot syndrome.
Overall, Dasari and colleagues concluded that “Fruquintinib treatment resulted in a significant and clinically meaningful benefit in overall survival compared with placebo in patients with refractory metastatic colorectal cancer.”
Read More Research on Colorectal and Gastrointestinal Cancers






 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.