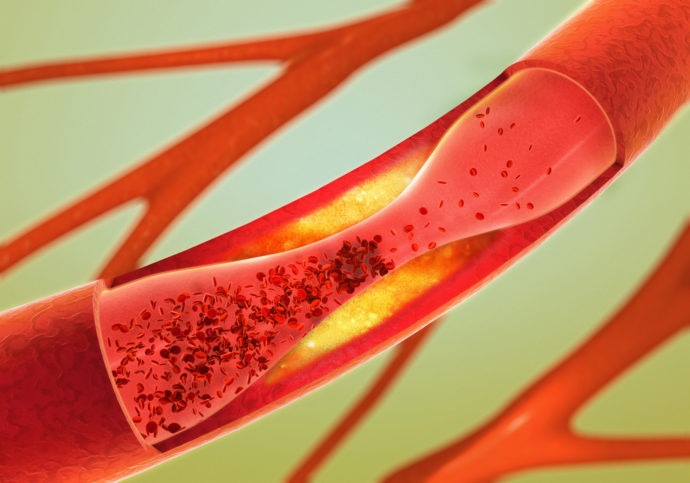
Icosapent ethyl was effective at slowing coronary atherosclerosis, according to new study results presented at the just-wrapped American Heart Association 2019 Scientific Sessions.
The Effect Of Icosapent Ethyl On Progression Of Coronary Atherosclerosis In Patients With Elevated Triglycerides On Statin Therapy (EVAPORATE) was a randomized, double-blind study designed to evaluate the efficacy of a treatment regimen of 4 grams per day of icosapent ethyl added onto background statin therapy in statin-treated patients with atherosclerosis. The researchers included 80 patients in the trial, who had atherosclerosis as measured by MDCT, with baseline low-density lipoprotein cholesterol (LDL-C) 40 to 115 mg/dL and persistent high triglyceride levels of 200 to 499 mg/dL. Patients underwent scans at nine months, and were followed-up with MDCT at zero, nine, and 18 months. The primary study endpoint was a change in low-attenuation plaque volume, with secondary endpoints of incident plaque rates (quantitative changes in different plaque types and morphology), changes in inflammation markers, changes in lipids and lipoproteins, and the relationship between changes with noncalcified coronary plaque burden or plaque-vulnerability features.
According to the study results, at nine months, the prespecified interim analysis showed that icosapent ethyl slowed progression in low-attenuation plaque (21%, P=0.469), non-calcified plaque (19%, P=0.010), total plaque (P=0.0004), fibrous plaque (P=0.011), calcified plaque (89%, P=0.001), and an increase in fibrofatty plaque (P=0.650). The efficacy was consistent across multiple subgroups, including in those with baseline triglycerides from 135-499 mg/dL.
“EVAPORATE [was] the first study using MDCT to evaluate the effects of icosapent ethyl as an adjunct to statin therapy on plaque characteristics in a high CV risk American population with persistent high triglyceride levels and will assess whether these effects correlate with lipid changes and inflammatory markers, the study abstract indicated. “EVAPORATE may provide important mechanistic data that may have relevance to the REDUCE-IT results and clinical use of icosapent ethyl.”





 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.